The mass of neutrons and protons, is approximately equal. It contains positively charged particles called protons, and non-electrical charge particles, called neutrons. Figure 2.4 "Rutherford’s Metal-Foil Experiments" shows how this model explains the experimental results. The nucleus is in the central part of the atom. Rutherford called his description the “planetary model” of the atom. As a result, an atom consists largely of empty space.

Most of the mass of an atom is in the nucleus, while the orbiting electrons account for an atom’s size. Electrons are outside the nucleus and orbit about it because they are attracted to the positive charge in the nucleus. On Earth, a typical atom has the same number of electrons as protons, and these electrons follow complex orbital patterns. For example, any atom with six protons is the element carbon, with eight protons is oxygen, with 26 is iron, and with 92 is uranium. Protons and neutrons are concentrated in a central region he called the nucleus The central part of an atom that contains protons and neutrons. The type of element is determined by the number of protons in the nucleus of the atom. Rutherford proposed the following model to explain these experimental results. Rutherford once said, “It was almost as incredible as if you fired a 15-inch shell at a piece of tissue paper and it came back and hit you.” Each atom is made up of three subatomic particles: protons, neutrons, and electrons. Some were even deflected back toward the source. All matter is composed of atoms, the smallest individual units of elements. Most of the particles traveled straight through the foil, but some alpha particles were deflected off to one side. Table 2.4 "Properties of the Subatomic Particles" lists some of their important characteristics and the symbols used to represent each particle.įigure 2.3 The Geiger-Marsden Experimental SetupĮxperiments using this setup were used to investigate the structure of atoms. We understand now that all atoms can be broken down into subatomic particles: protons, neutrons, and electrons. Evidence produced in 1932 established the existence of the neutron A subatomic particle with no electric charge., a particle with about the same mass as a proton but with no electrical charge. Because opposite charges attract each other (while like charges repel each other), protons attract electrons (and vice versa).įinally, additional experiments pointed to the existence of a third particle. Although still incredibly small, the mass of a proton is 1.673 × 10 −27 kg, which is almost 2,000 times greater than the mass of an electron.
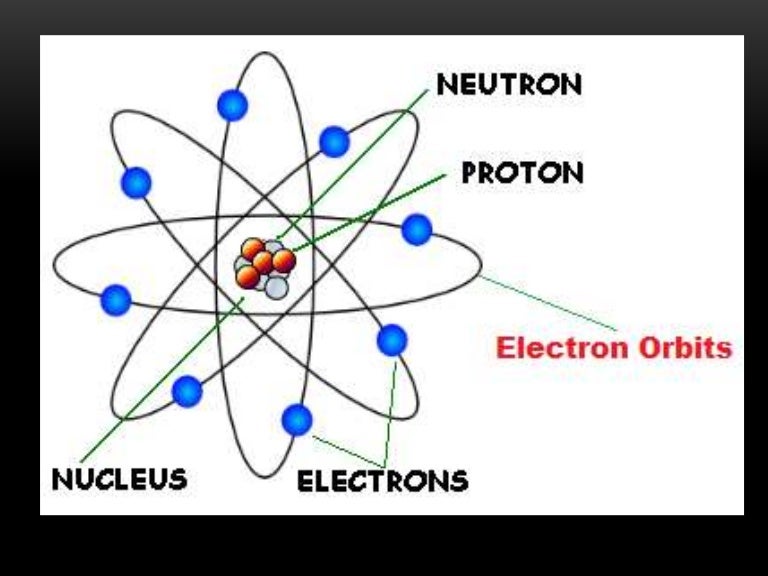
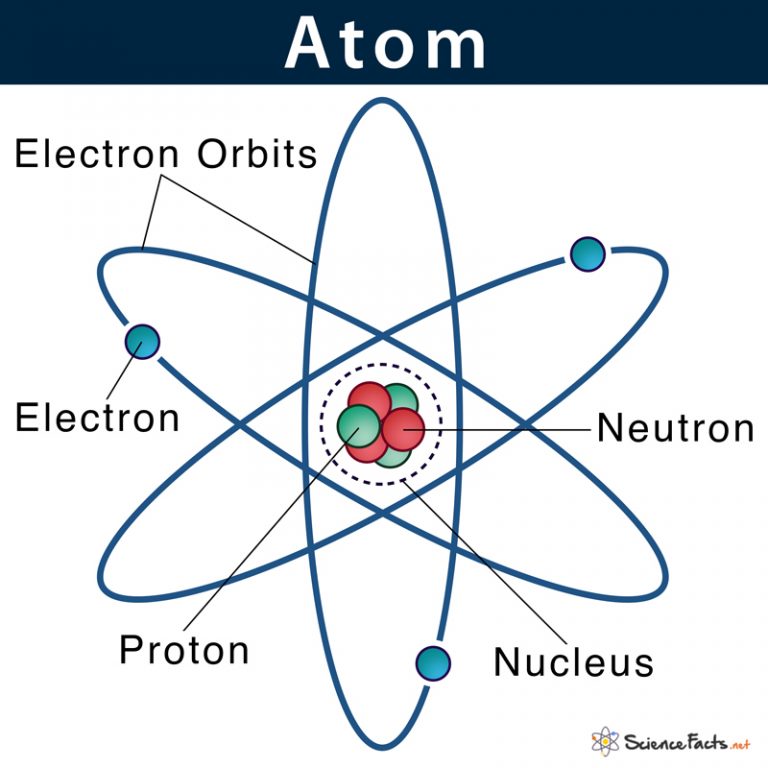
Another major difference between a proton and an electron is mass. has the same amount of charge as an electron, but its charge is positive, not negative. A proton A subatomic particle with a positive charge. Experiments with magnetic fields showed that the electron has a negative electrical charge.īy 1920, experimental evidence indicated the existence of a second particle. It is an extremely tiny particle, with a mass of about 9.109 × 10 −31 kg. The first subatomic particle was identified in 1897 and called the electron A subatomic particle with a negative electric charge.

